 Cranial Electrotherapy Stimulation (CES) is the FDA name for devices which send micro currents of electricity across patient’s cranium. FDA has cleared the CES devices for treatment of anxiety and insomnia and (with PMA) for treatment of depression. The current level delivered across patient’s brain is limited to under 1.5mA which is several orders of magnitude lower than in treatments like ECT.
Cranial Electrotherapy Stimulation (CES) is the FDA name for devices which send micro currents of electricity across patient’s cranium. FDA has cleared the CES devices for treatment of anxiety and insomnia and (with PMA) for treatment of depression. The current level delivered across patient’s brain is limited to under 1.5mA which is several orders of magnitude lower than in treatments like ECT.
The history of CES technology dates back to 1950s, when the scientists were investigating the effects of micro-current stimulation on the ability to promote sleep. This was done during the space program days when the astronauts were exhibiting severe anxiety and insomnia during the pioneering days of space exploration. Given that an astronaut’s headgear is already instrumented with various sensors and electrodes, the space program engineers cleverly took advantage of this, delivering micro-currents of electricity to astronaut’s brain. The effects were surprisingly positive. The astronauts were getting relaxed; some of them so much so that they were falling asleep inside the space simulators! Hence, the first clinical indication for CES technology centered around anxiety reduction and, often comorbid, insomnia.
In the United States, the first micro-current CES device achieving FDA approval in 1978 was called Neurotone 101. Since then, over a hundred of clinical trials were conducted demonstrating the safety and efficacy of CES for treatment of anxiety, depression, and insomnia.
Additionally, over the last few decades, Cranial Electrotherapy Stimulators have been extensively studied for treatment of variety of disorders. The following table summarizes a meta-analysis of number of clinical trials for each disorder*:
| Study Subject | Number of Studies | Number of Subjects |
Average Improvement |
| Insomnia |
18 |
648 | 62% |
| Depression |
18 |
853 |
47% |
| Anxiety |
38 |
1,496 |
58% |
| Drug Abstinence |
15 |
535 |
60% |
|
Cognitive Dysfunction |
13 | 648 |
44% |
*Source: Ray B. Smith, Ph.D. Cranial Electrotherapy Stimulation, 17, 2007.
The paper by Barclay TH, Barclay RD. A clinical trial of cranial electrotherapy stimulation for anxiety and comorbid depression, Journal of Affective Disorders, 164:171-177, 2014, presented at the American Psychological Association National Conference, Honolulu, HI, July 2013, represents one of the recent clinical trials with a reasonable large sample size (N=115) for patients exhibiting anxiety with comorbid depression. The authors concluded that, “CES significantly decreases anxiety and comorbid depression. Subjects reported no adverse events during the study.”
A meta analysis of the “18 most carefully conducted randomized controlled trials of CES vs sham” revealed that, “CES to be significantly more effective than sham treatment (p<0.05).” Please see the abstract of the study here: Meta-analysis of randomized controlled trials of cranial electrostimulation. Efficacy in treating selected psychological and physiological conditions.
DeFelice (1997) conducted a meta analysis of 34 controlled clinical trials concerning the efficacy of CES in the treatment of anxiety and other stress‐related disorders were evaluated. The author states, “Overall the results suggest that CES may be capable of producing significant (p<0.05) benefit in the short‐term symptomatic relief of anxiety and other stress‐related disorders. CES may be effective alone and as an adjunct to other conservative measures of treatment. The primary mechanism of action of CES appears to be a direct effect on the brain followed by secondary responses.”
Cervella Efficacy
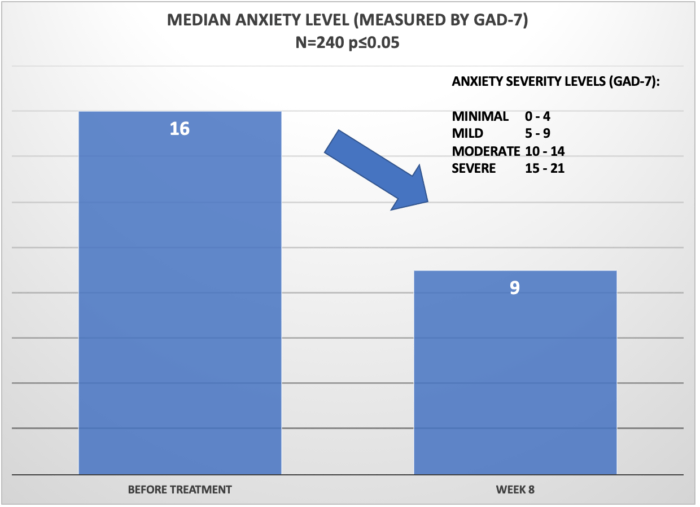
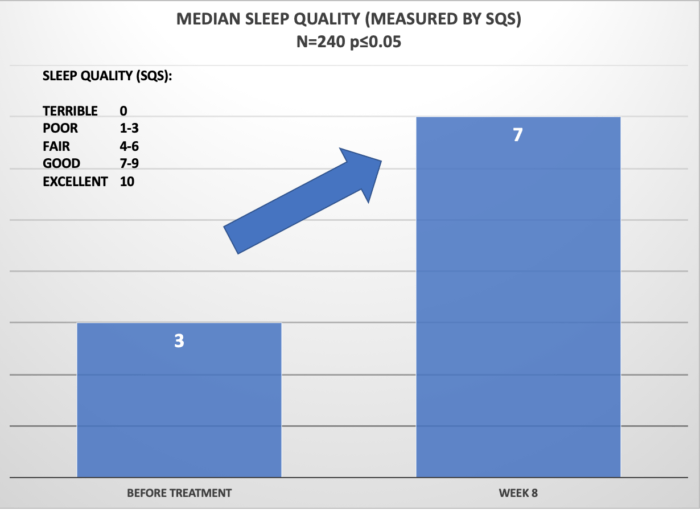
Below, is the summary of the results of clinical retrospective study on N=240 patients using Cervella for 8 weeks. The data was analyzed for significance level p<=0.05 and power 80%. Patients were instructed to use Cervella at the default settings (30 min duration, frequency of 100Hz) daily and adjust the treatment intensity so that the “tingling” sensation was barely perceptible.
Success in reduction of anxiety symptoms was defined as reduction of 5 or more points on the GAD-7. This corresponds to dropping in the severity level by a severity category (e.g. improvement from Severe to Moderate on the GAD-7 score).
For insomnia reduction, the success factor was defined by improvement of sleep quality as measured by the SQS by 3 or more points on the SQS scale which also corresponds to “category” improvement in terms of sleep quality (e.g. improvement from Poor sleep to Fair sleep quality).
Overall, 92% of patients reported that the Cervella was beneficial with treatment of the underlying conditions as defined by the success criteria defined above. This is represented as a statistically-significant reduction of the conditions as evaluated by the GAD-7 (Anxiety) or SQS (Insomnia).
Mechanisms of Action

According to clinical studies, there are several mechanisms of actions for the Cranial Electrotherapy Stimulator. One of the mechanisms is changes in cortical brain activity, including effects on the DMN (Default Mode Network). According to Feusner (2012) study, “Results suggest that CES causes cortical brain deactivation, with a similar pattern for high- and low-frequency stimulation, and alters connectivity in the DMN.”
Additional studies by Kennerly (2004) suggest that the CES stimulation affects the endogenous brain oscillations (specifically increase in alpha activity and decrease in delta activity).
Liss (1996) postulates that the CES affects the levels of neurotransmitter levels in the CSF, specifically serotonin and beta-endorphin which may also contribute to reduction of patient’s depressive mood and increase of the sense of well-being.
Clinical Research
The following is a partial list of clinical trials and research on the efficacy of Cranial Electrotherapy Stimulation. Note that, as per the FDA requirements, Cervella is substantially equivalent in the underlying principle of operation (current intensity levels, frequencies, treatment duration) to the CES devices cited in the various clinical trials.
- Barclay TH, Barclay RD. A clinical trial of cranial electrotherapy stimulation for anxiety and comorbid depression. Journal of Affective Disorders, 164:171-177, 2014.
- Klawansky S, Yeung A, Berkey C, Shah N, Phan H, Chalmers TC., Department of Health Policy and Management, Harvard School of Public Health, Boston, Massachusetts 02115, USA. Meta-analysis of randomized controlled trials of cranial electrostimulation. Efficacy in treating selected psychological and physiological conditions. J Nerv Ment Dis. 1997 Dec; 185 (12): 766-7
- Bystritsky A, Kerwin L, Feusner J.Department of Psychiatry and Biobehavioral Sciences, University of California, Los Angeles, CA 90095, USA. A Pilot Study of Cranial Electrotherapy Stimulation for Generalized Anxiety Disorder
- Jamie D. Feusner, Sarah Madsen, Teena D. Moody, Cara Bohon, Emily Hembacher, Susan Y. Bookheimer & Alexander Bystritsky. Department of Psychiatry and Biobehavioral Sciences, David Geffen School of Medicine, University of California, Los Angeles, California. Effects of cranial electrotherapy stimulation on resting state brain activity
- DeFelice EA. Cranial electrotherapy stimulation (CES) in the treatment of anxiety and other stress-related disorders: a review of controlled clinical trials. Stress Med. 1997;13:31–42.
Product Advantages 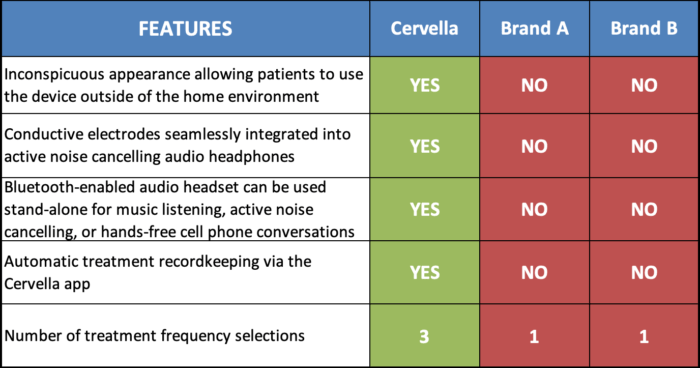
Cervella is the first CES device specifically developed for active-lifestyle patients who suffer from anxiety, depression, and insomnia disorders. Through patient research, we have found that many anxiety sufferers, as well as depression patients, are usually very self-conscious about their disorder. Cervella offers discreet features that allow patients to administer a treatment session at school, at work or even around town as needed. It also features Bluetooth sound integration with high quality stereo headphones, which adds to the great value and functionality of the device. The affordable price point keeps Cervella within reach of many patients. Insurance coverage, although not universal, is gradually increasing with several large national insurance providers. Our staff can assist you and your patient with the insurance reimbursement process.
Benefits for the Clinician
Because of the inconspicuous appearance of the Cervella device, patients are more apt to use Cervella outside of the comfort of their homes increasing the compliance and use of the device during stressful events. Furthermore, Cervella is the first and only CES device that is managed through an App on a smart device.
This feature allows for automated treatment data recording and sharing treatment history with patient’s provider via data export within the Cervella app in order to better manage the treatment plan.
Set-up and Operation Video
Please see the set-up and operation video for the Cervella Cranial Electrotherapy Stimulator below:
Patient Brochure
Please click on the button below to download a Patient Brochure in PDF format that you can share via e-mail with your patients.
Cervella Patient BrochureClinician FAQ
-
Q: How does Cervella work?
-
Q: Is Cervella FDA-approved?
-
Q: Can Cervella be used in conjunction with drug-based therapies or cognitive based therapies?
-
Q: What are the potential side-effects for Cervella?
-
Q: Who should not use Cervella?
-
Q: What is the recommended treatment protocol for the Cervella?
-
Q: What is the best time to use Cervella?
