Cervella Anxiety Insomnia, and Depression Long-Term Patient Study Results
In 2020, our team completed a clinical evaluation of 240 randomly-selected Cervella patients (57% female and 43% male) who were prescribed the Cervella medical device for treatment of anxiety, insomnia, depression, or combination of these three disorders. Each patient was evaluated before starting treatment and after approximately 8 weeks / 60 days from start of treatment. Patients were instructed to use the device daily for 30 minutes at the default 100Hz setting. Patients were free to choose the best time of day to use the treatment and the intensity of the treatment was also controlled by patient (since it varies from patient to patient) but all patients were instructed to only adjust the treatment intensity to the level where the “tingling” sensation is barely perceptible.
We are very happy to provide a summary of our results as the they have, frankly, exceeded our expectations. Over 92% of patients reported that the Cervella device had positive effect on improving their condition. Below, we would like to provide more detailed results of the year-long study.
First, let us explain the methodology used to score the level of pre and post-treatment disorders. Patients who have been diagnosed with Anxiety, were evaluated using GAD-7 (General Anxiety Disorder-7) questionnaire which is an industry standard for evaluating anxiety level. Patients who have been diagnosed with Insomnia, were evaluated with SQS (Sleep Quality Scale). Patients who were diagnosed with Depression were evaluated with PHQ-9 (Patient Health Questionnaire-9).
The data was analyzed for significance level p<=0.05 and power >=80%.
It is important to note that, with respect to anxiety and insomnia, and depression, patients are frequently diagnosed with a combination of these disorders due to relatively frequent amount of comorbidity between these disorders. For example, patients suffering from anxiety are often insomniac and vice-versa. Patients who have been diagnosed with more than one of these disorders were evaluated using measures specifically applicable to each disorder separately. For example, patients who were diagnosed and prescribed the Cervella device for anxiety and insomnia, were evaluated using GAD-7 for anxiety and they were also asked to complete the SQS for insomnia.
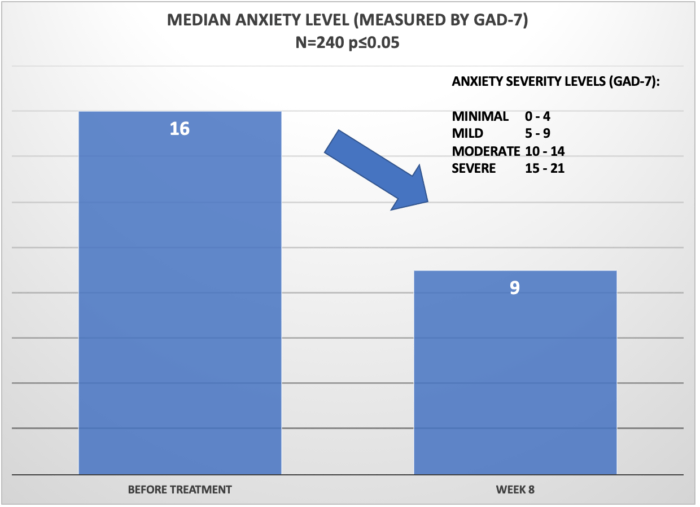
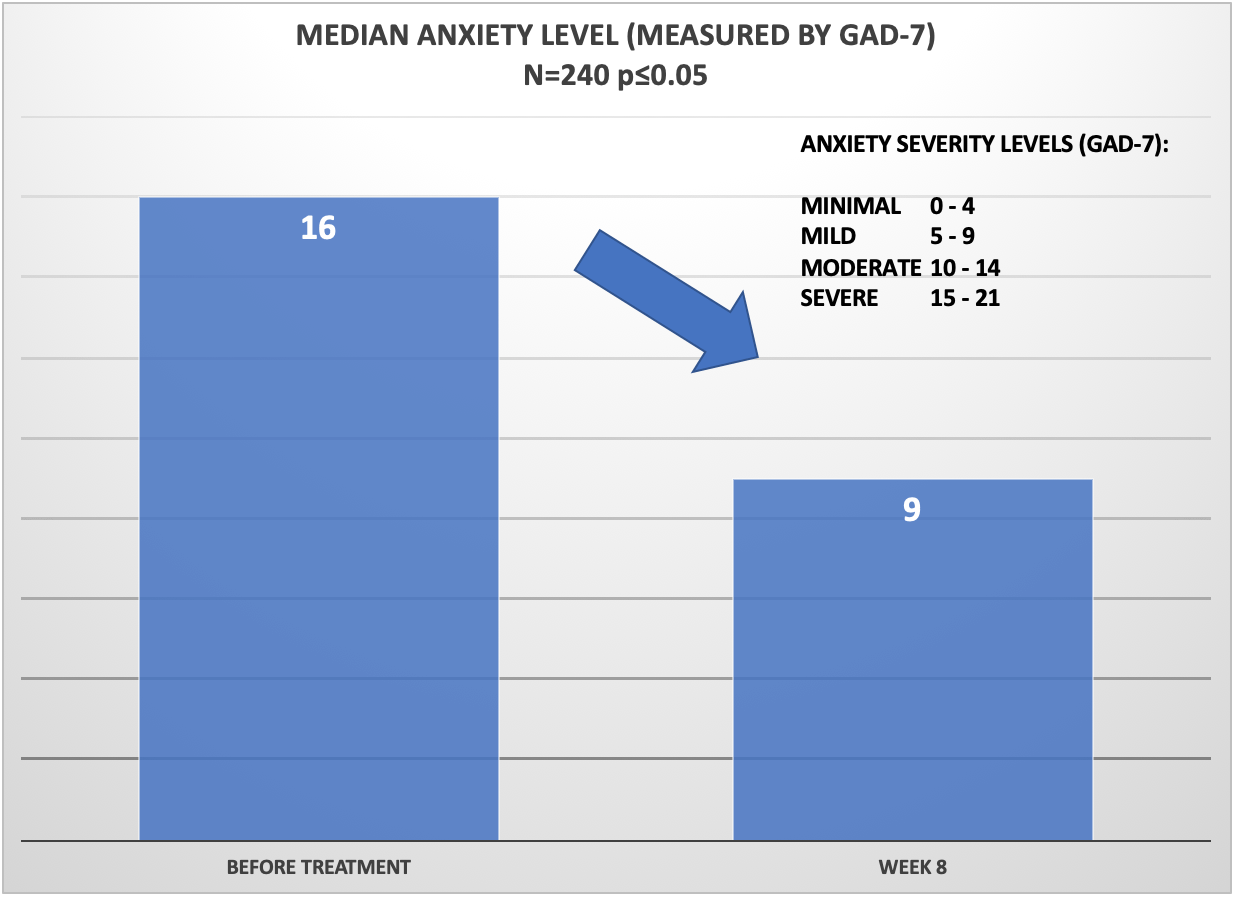
This graph shows the reduction of median anxiety level, as measured by GAD-7). The median anxiety level before treatment was 16 which is in the 16 – “Severe” anxiety category. Post treatment (approx. 8 weeks) the median anxiety level among patent population was reported as 9 – “Mild”, a reduction of 7 points on the GAD-7 scale.
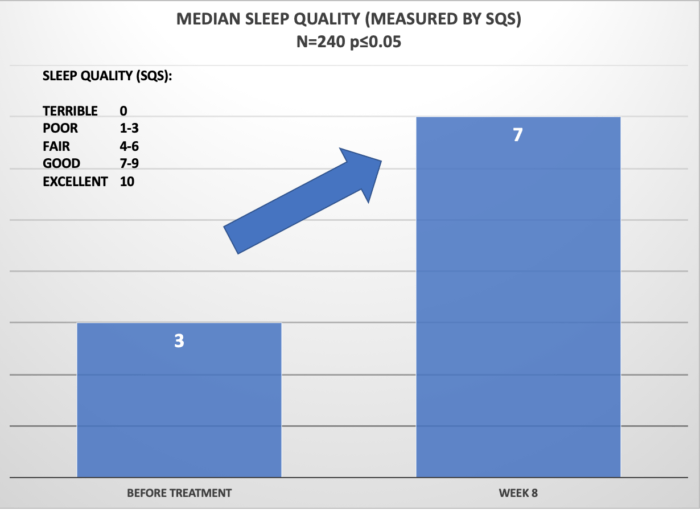
The Insomnia reduction, or sleep quality improvement is also quite remarkable. Before treatment, the median sleep quality was reported as 3 – “Poor.” After approximately 60 days/8 weeks, the medial sleep quality improved to 7 – “Good” which is a 4-point improvement on the SQS scale.
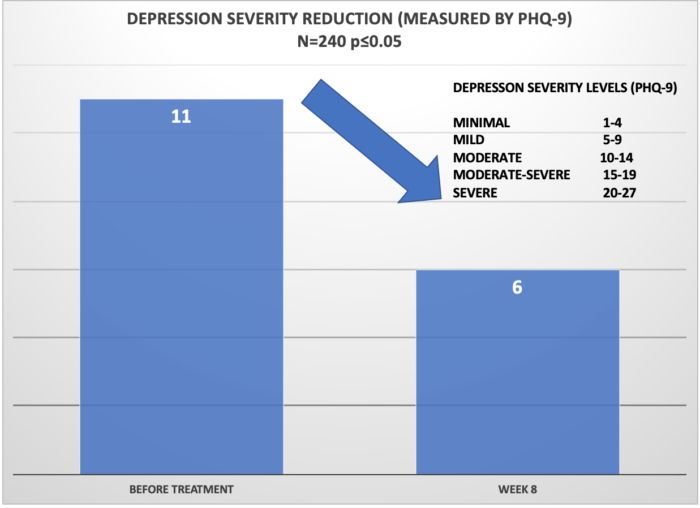
The study also found measurable and statistically-significant depression severity reduction as measured by Patient Health Questionnaire-9. At the onset of the treatment, the median depression severity level was reported as 11 – “Moderate.” After 8 weeks, the median depression level was reported as 6 – “Mild.” Note that out of the three disorders studied (anxiety, depression, and insomnia), depression is most difficult to treat and the reduction of depression is often slower and more gradual than with the other two disorders.
There were no serious adverse events reported and only minor adverse events were reported (<1% of patients). Most common minor adverse event were dizziness/headaches (which typically is resultant of using the device at too high than comfortable current level settings). We also had one patient who was allergic to the conductive gel (which is hypoallergenic and FDA-approved). For that patient, water was successfully used as a substitute for the gel and the patient completed the treatment successfully.
Given the overall 92% positive response rate, statistically-significant improvements in anxiety reduction and sleep quality, and the very low instances of non-serious adverse events and no serious adverse events, we can conclude that the Cervella is both safe and effective for treatment of Anxiety and Insomnia, Depression, or comorbidity of these disorders.
